First up, a pair of researchers from the University of Arizona - this is a segment that aired on Arizona Public Media (NPR, PBS) - discuss the research their team conducted to bring a new understanding to how axons and dendrites are created.
UA Researchers Discover How Nerve Cells Make Connections
Story by Mary Olivas
October 1, 2013
University of Arizona researchers have made a discovery about how nerve cells make connections. This discovery is an important step towards curing conditions like spinal cord injuries and Alzheimer's.
Developed nerves have a short, truncated branch called an axon and long branch called a dendrite.
Sourav Ghosh, UA assistant professor of cellular and molecular medicine, said that this structure determines how nerves send signals.
"When nerves fire and communicate information, it's direction," Ghosh said. "It's sort of the basis for directionality of function."
Ghosh and UA doctoral candidate Sara Parker have been studying the polarity of neurons in their lab by closely examining a family of proteins and watching the neurons go through their development process.
Parker said she would knock down the levels of a protein or introduce excessive amounts of the protein to try and influence the establishment of a neuron's polarity.
"What we found is that when I disrupt this short form, I end up creating neurons developing multiple axons," Parker explained.
She added that usually neurons only have one axon.
This new understanding of how axons and dendrites are created could possibly help those with spinal injuries or neurodegenerative diseases.
"In traumatic injury what happens is that the axon is severed, but the cell is still alive and if we understand this mechanism and play with it to make the cell come up with a new axon, that would be exciting," Ghosh said.
Here is a more in-depth report on this study from Science Daily:
The full article is behind a paywall, but here is the full citation:
How Neurons Get Wired
Aug. 14, 2013 — Two different versions of the same signaling protein tell a nerve cell which end is which, UA researchers have discovered. The findings could help improve therapies for spinal injuries and neurodegenerative diseases.
University of Arizona scientists have discovered an unknown mechanism that establishes polarity in developing nerve cells. Understanding how nerve cells make connections is an important step in developing cures for nerve damage resulting from spinal cord injuries or neurodegenerative diseases such as Alzheimer's.
In a study published on Aug. 12 in the journal Proceedings of the National Academy of Sciences, UA doctoral student Sara Parker and her adviser, assistant professor of cellular and molecular medicine Sourav Ghosh, report that the decision which will be the "plus" and the "minus" end in a newborn nerve cell is made by a long and a short version of the same signaling molecule.
Nerve cells -- or neurons -- differ from many other cells by their highly asymmetric shape: Vaguely resembling a tree, a neuron has one long, trunk-like extension ending in a tuft of root-like bristles. This is called the axon. From the opposite end of the cell body sprout branch-like structures known as dendrites. By connecting the "branches" of their dendrites to the "root tips" of other neurons' axons, nerve cells form networks, which can be as simple as the few connections involved in the knee-jerk reflex or as complex as those in the human brain.
Parker and her team found that embryonic nerve cells manufacture a well-known signaling enzyme called Atypical Protein Kinase C (aPKC) in two varieties: a full-length one and a truncated one. Both varieties compete to bind the same molecular partner, a protein called Par3. If the short form of aPKC pairs up with Par3, it tells the cell to grow a dendrite, and if the long one pairs up with Par3, it will make an axon instead.
When the researchers blocked the production of the short form, the nerve cell grew multiple axons and no dendrites. When they created an artificial abundance of the short form, dendrites formed at the expense of axons. UA undergraduate student Sophie Hapak performed many of the experiments revealing how the two isoforms compete for Par3.
"We show that wiring a neuronal circuit is much more complex than previously thought," said Ghosh. "The process has a built-in robustness that explicitly defines which part of the cell is 'positive' and which is 'negative.'"
"In order to have a functioning neuronal circuit, you have to have receiving and sending ends," Parker said. "Initially, when a neuron is formed, it lacks the polarity it needs once it develops into a part of a circuit. The mechanism we discovered establishes that polarity."
"How the various brain regions are wired is the basis of emotion, memory and all cognitive functions," said Ghosh, who is a member of the UA's BIO5 Institute. "Establishing neuronal polarity in single neurons is absolutely essential for neuronal circuits to form."
"If we understand this mechanism, we could think about methods to spur new axons after the original ones were severed in a traumatic spinal cord injury, for example," Ghosh said.
The findings defy conventional wisdom, which maintains that a developing neuron will make dendrites by default unless instructed by the long form of aPKC to make an axon instead. By cultivating and studying neurons just after they formed, Parker and her group found that both forms of aPKC, long and short, are initially distributed equally throughout the cell. These forms subsequently segregate into different parts of the cell as the neuron matures and establishes polarity.
Because the cells were isolated from rat brains and kept in culture, the researchers could demonstrate that no external clues from other cells are needed to instruct a developing neuron. Whether the establishment of polarity is a random process or whether other signals yet to be identified play a role in regulating the abundance of the two aPKC varieties is not known.
Funded by the National Institutes of Health and a scholarship awarded to Parker by the Achievement Rewards for College Scientists Foundation, Inc., this research resulted from a collaboration of three UA departments -- cellular and molecular medicine, pharmacology and physiology.
S.S. Parker, E.K. Mandell, S.M. Hapak, I.Y. Maskaykina, Y. Kusne, J.-Y. Kim, J.K. Moy, P.A. St. John, J.M. Wilson, K.M. Gothard, T.J. Price, S. Ghosh. Competing molecular interactions of aPKC isoforms regulate neuronal polarity. Proceedings of the National Academy of Sciences, 2013; DOI: 10.1073/pnas.1301588110
* * * * *
The second article looks at how synapses are formed - the original article is open access and will be partially presented below. Bur first, here is a summary press release posted at Science Codex.
The full article is open access and appeared at PLoS Computational Biology - below is the abstract, author's summary, and the full citation. Follow the title link to read the whole article.New theory of synapse formation in the brain
Jülich, 10 October 2013 – The human brain keeps changing throughout a person's lifetime. New connections are continually created while synapses that are no longer in use degenerate. To date, little is known about the mechanisms behind these processes. Jülich neuroinformatician Dr. Markus Butz has now been able to ascribe the formation of new neural networks in the visual cortex to a simple homeostatic rule that is also the basis of many other self-regulating processes in nature. With this explanation, he and his colleague Dr. Arjen van Ooyen from Amsterdam also provide a new theory on the plasticity of the brain – and a novel approach to understanding learning processes and treating brain injuries and diseases.
The brains of adult humans are by no means hard wired. Scientists have repeatedly established this fact over the last few years using different imaging techniques. This so-called neuroplasticity not only plays a key role in learning processes, it also enables the brain to recover from injuries and compensate for the loss of functions. Researchers only recently found out that even in the adult brain, not only do existing synapses adapt to new circumstances, but new connections are constantly formed and reorganized. However, it was not yet known how these natural rearrangement processes are controlled in the brain. In the open-access journal PLOS Computational Biology, Butz and van Ooyen now present a simple rule that explains how these new networks of neurons are formed (DOI: 10.1371/journal.pcbi.1003259).
"It's very likely that the structural plasticity of the brain is the basis for long-term memory formation," says Markus Butz, who has been working at the recently established Simulation Laboratory Neuroscience at the Jülich Supercomputing Centre for the past few months. "And it's not just about learning. Following the amputation of extremities, brain injury, the onset of neurodegenerative diseases, and strokes, huge numbers of new synapses are formed in order to adapt the brain to the lasting changes in the patterns of incoming stimuli."
Activity regulates synapse formation
These results show that the formation of new synapses is driven by the tendency of neurons to maintain a 'pre-set' electrical activity level. If the average electric activity falls below a certain threshold, the neurons begin to actively build new contact points. These are the basis for new synapses that deliver additional input – the neuron firing rate increases. This also works the other way round: as soon as the activity level exceeds an upper limit, the number of synaptic connections is reduced to prevent any overexcitation – the neuron firing rate falls. Similar forms of homeostasis frequently occur in nature, for example in the regulation of body temperature and blood sugar levels.
However, Markus Butz stresses that this does not work without a certain minimal excitation of the neurons: "A neuron that no longer receives any stimuli loses even more synapses and will die off after some time. We must take this restriction into account if we want the results of our simulations to agree with observations." Using the visual cortex as an example, the neuroscientists have studied the principles according to which neurons form new connections and abandon existing synapses. In this region of the brain, about 10 % of the synapses are continuously regenerated. When the retina is damaged, this percentage increases even further. Using computer simulations, the authors succeeded in reconstructing the reorganization of the neurons in a way that conforms to experimental results from the visual cortex of mice and monkeys with damaged retinas.
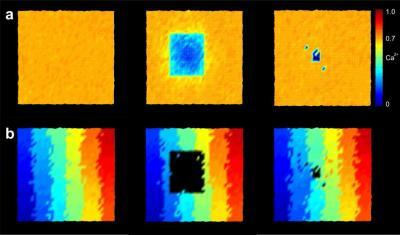 (Photo Credit: Source: PLOS Computational Biology)
(Photo Credit: Source: PLOS Computational Biology)
Reorganization in the visual cortex: before (left) and just after damage to the retina (centre), and in a later phase (right). Most of the neurons in the zone where images are projected from the damaged retina can achieve their original activity level again thanks to new connections formed with neighboring cells. The colors in the lower part of the image indicate the points on the retina from which the input to which the neurons show the strongest reaction is received.
The visual cortex is particularly suitable for demonstrating the new growth rule, because it has a property referred to as retinotopy: This means that points projected beside each other onto the retina are also arranged beside each other when they are projected onto the visual cortex, just like on a map. If areas of the retina are damaged, the cells onto which the associated images are projected receive different inputs. "In our simulations, you can see that areas which no longer receive any input from the retina start to build crosslinks, which allow them to receive more signals from their neighbouring cells," says Markus Butz. These crosslinks are formed slowly from the edge of the damaged area towards the centre, in a process resembling the healing of a wound, until the original activity level is more or less restored.
Synaptic and structural plasticity
"The new growth rule provides structural plasticity with a principle that is almost as simple as that of synaptic plasticity," says co-author Arjen van Ooyen, who has been working on models for the development of neural networks for decades. As early as 1949, psychology professor Donald Olding Hebb discovered that connections between neurons that are frequently activated will become stronger. Those that exchange little information will become weaker. Today, many scientists believe that this Hebbian principle plays a central role in learning and memory processes. While synaptic plasticity in involved primarily in short-term processes that take from a few milliseconds to several hours, structural plasticity extends over longer time scales, from several days to months.
Structural plasticity therefore plays a particularly important part during the (early) rehabilitation phase of patients affected by neurological diseases, which also lasts for weeks and months. The vision driving the project is that valuable ideas for the treatment of stroke patients could result from accurate predictions of synapse formation. If doctors knew how the brain structure of a patient will change and reorganize during treatment, they could determine the ideal times for phases of stimulation and rest, thus improving treatment efficiency.
New approach for numerous applications
"It was previously assumed that structural plasticity also follows the principle of Hebbian plasticity. The findings suggest that structural plasticity is governed by the homeostatic principle instead, which was not taken into consideration before," says Prof. Abigail Morrison, head of the Simulation Laboratory Neuroscience at Jülich. Her team is already integrating the new rule into the freely accessible simulation software NEST, which is used by numerous scientists worldwide.
These findings are also of relevance for the Human Brain Project. Neuroscientists, medical scientists, computer scientists, physicists, and mathematicians in Europe are working hand in hand to simulate the entire human brain on high-performance computers of the next generation in order to better understand how it functions. "Due to the complex synaptic circuitry in the human brain, it's not plausible that its fault tolerance and flexibility are achieved based on static connection rules. Models are therefore required for a self-organization process," says Prof. Markus Diesmann from Jülich's Institute of Neuroscience and Medicine, who is involved in the project. He heads Computational and Systems Neuroscience (INM-6), a subinstitute working at the interface between neuroscientific research and simulation technology.
Source: Forschungszentrum Juelich
A Simple Rule for Dendritic Spine and Axonal Bouton Formation Can Account for Cortical Reorganization after Focal Retinal Lesions
Markus Butz, Arjen van Ooyen
Abstract
Lasting alterations in sensory input trigger massive structural and functional adaptations in cortical networks. The principles governing these experience-dependent changes are, however, poorly understood. Here, we examine whether a simple rule based on the neurons' need for homeostasis in electrical activity may serve as driving force for cortical reorganization. According to this rule, a neuron creates new spines and boutons when its level of electrical activity is below a homeostatic set-point and decreases the number of spines and boutons when its activity exceeds this set-point. In addition, neurons need a minimum level of activity to form spines and boutons. Spine and bouton formation depends solely on the neuron's own activity level, and synapses are formed by merging spines and boutons independently of activity. Using a novel computational model, we show that this simple growth rule produces neuron and network changes as observed in the visual cortex after focal retinal lesions. In the model, as in the cortex, the turnover of dendritic spines was increased strongest in the center of the lesion projection zone, while axonal boutons displayed a marked overshoot followed by pruning. Moreover, the decrease in external input was compensated for by the formation of new horizontal connections, which caused a retinotopic remapping. Homeostatic regulation may provide a unifying framework for understanding cortical reorganization, including network repair in degenerative diseases or following focal stroke.
Author Summary
The adult brain is less hard-wired than traditionally thought. About ten percent of synapses in the mature visual cortex is continually replaced by new ones (structural plasticity). This percentage greatly increases after lasting changes in visual input. Due to the topographically organized nerve connections from the retina in the eye to the primary visual cortex in the brain, a small circumscribed lesion in the retina leads to a defined area in the cortex that is deprived of input. Recent experimental studies have revealed that axonal sprouting and dendritic spine turnover are massively increased in and around the cortical area that is deprived of input. However, the driving forces for this structural plasticity remain unclear. Using a novel computational model, we examine whether the need for activity homeostasis of individual neurons may drive cortical reorganization after lasting changes in input activity. We show that homeostatic growth rules indeed give rise to structural and functional reorganization of neuronal networks similar to the cortical reorganization observed experimentally. Understanding the principles of structural plasticity may eventually lead to novel treatment strategies for stimulating functional reorganization after brain damage and neurodegeneration.
Full Citation:
Butz M, van Ooyen A. (2013, Oct 10). A Simple Rule for Dendritic Spine and Axonal Bouton Formation Can Account for Cortical Reorganization after Focal Retinal Lesions. PLoS Comput Biol 9(10): e1003259. doi:10.1371/journal.pcbi.1003259

No comments:
Post a Comment